|
The law of conservation of mass and constant proportions are the basis that helps explain Dalton’s atomic theory. The definition of dalton's atomic theory brought the novel concept of calculating relative atomic weights. Although connections of his work have been made with several other chemists of the time. Dalton’s idea for the theory is believed to be inspired by the physical properties of gases. The theory of Dalton was published in the paper “New Chemical Philosophy”. However, the ideas from researches of methane and ethylene might have helped define Dalton’s atomic theory at the time. Dalton’s model came almost two millennia later and brought further light to the topic. Democritus was known to be the first to suggest that matter is made up of particles. 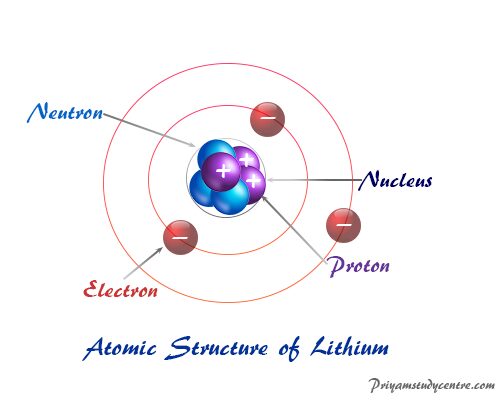
The matter has been a subject of fascination since the beginning. (Image to be added soon) Dalton’s Atomic Model Each atom was circular and bears different symbols. Dalton’s model suggested the atom to be a ball-like structure that cannot be further divided. He considered the atom as the smallest, indivisible unit of matter and wrote several postulates. 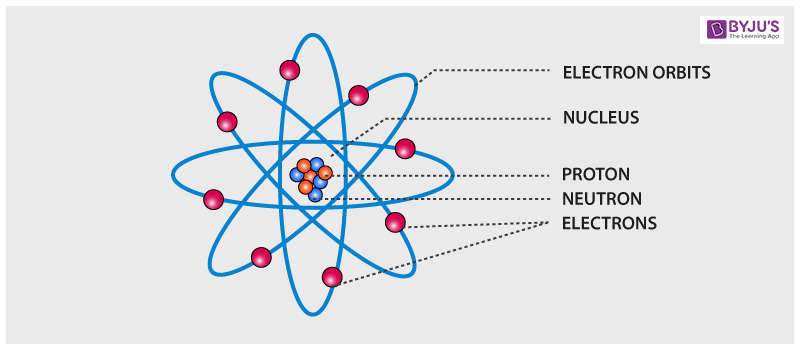
Dalton's atomic model showed the way to many future works, researches regarding atomic theory, even though his conclusions were rather incorrect. He was a British physicist, chemist, and meteorologist who is well known for many of his contributions to the pioneering research of atoms, the law of partial pressures, Daltonism, etc. The introduction to the early theory of the atom was done by a scientist named John Dalton (1766-1844).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
